 The technology has regained popularity for off-the-grid applications where daily charging makes it an appropriate technology. Some examples are London underground electric locomotives and New York City Subway car – R62A. Many railway vehicles use NiFe batteries. Due to its low specific energy, poor charge retention, and high cost of manufacture, other types of rechargeable batteries have displaced the nickel–iron battery in most applications. 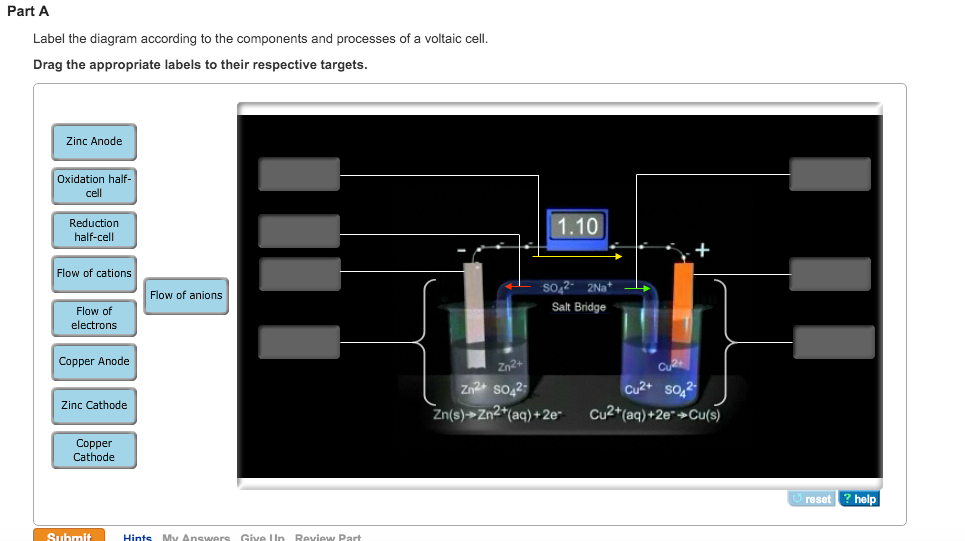
It is often used in backup situations where it can be continuously charged and can last for more than 20 years. It is a very robust battery which is tolerant of abuse, (overcharge, overdischarge, and short-circuiting) and can have very long life even if so treated. The active materials are held in nickel-plated steel tubes or perforated pockets. The nickel–iron battery (NiFe battery) is a rechargeable battery having nickel(III) oxide-hydroxide positive plates and iron negative plates, with an electrolyte of potassium hydroxide. A stack of three cells should generate enough voltage to light a red LED, which usually require around 1.7 volts.Thomas Edison in 1910 with his nickel-iron cell from his own production line The penny battery you created for this Snack has four cells. With a voltmeter, you can see that each cell can generate over 0.6 volts. In Volta’s battery and your penny battery, an oxidation reaction occurs at the zinc electrode that releases electrons and a reduction reaction occurs at the copper electrode that uses them. This is also called a voltaic pile, which is named after Alessandro Volta, who created the first battery in 1800 by alternating zinc and copper electrodes with sulfuric acid between them. By stacking additional matboards and sanded pennies, you’ve created a battery, which is a series of electrochemical cells. The matboard soaked in salty vinegar water serves as the electrolyte between the two terminals.Įach zinc-matboard-copper stack represents one individual cell. By sanding off one face of a penny, you create a zinc electrode that can pair with the copper electrode on the face of the next penny. Pennies that were made after 1982 have zinc cores that are plated with copper. When these electrodes are connected by a wire, electrons will move from one surface to the other, creating an electric current. When two different metals are connected by an electrolyte, a chemical reaction occurs at each metal surface, called electrodes, that either releases or uses electrons. 
The LED will grow fainter as the matboards dry out, but it should stay lit for 24 hours! To recharge, just re-soak the matboards and reassemble.īatteries are devices that convert chemical energy into electrical energy. If you want your LED to stay lit without having to hold it, wrap the entire assembly together with electrical tape. Did the LED turn on? If not, make sure the LED leads are oriented correctly and use a paper towel to wipe off any excess water from the penny-matboard stack.Ĭheck the voltage of the battery with a voltmeter by placing the leads on the top and bottom of your penny battery. Make sure that the leads don't touch any other layer. Touch the longer lead to the intact penny on the top and the shorter lead to the bottom of the stack. Test your battery by connecting your LED. Make sure the pennies aren't directly touching each other and, likewise, that the pieces of matboard aren't touching each other. When you're done, you should have alternating layers of pennies and matboard with all the zinc sides facing up, and both the very top and the very bottom of your stack (facing down) should be copper. Finally, place the unsanded penny at the very top. Take your next piece of matboard and place it on top, and continue stacking pennies and matboard on top of each other to make a neat pile. Then stack another sanded penny (again, zinc side facing up) on top of this. 
Take one of your sanded pennies with the zinc side facing up (copper side down), and place a damp piece of matboard on it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
